Do You Have To Take Beta Blockers After Valve Repair
Impact of beta blockers on patients undergoing transcatheter aortic valve replacement: the OCEAN-TAVI registry

Abstract
Objective In that location is paucity of data on optimal medical treatment, including use of beta blockers for patients undergoing transcatheter aortic valve replacement (TAVR). The report aimed to investigate the clan of beta blockers and clinical outcomes following TAVR.
Methods We examined data of 2563 patients who underwent TAVR between October 2022 and May 2022 obtained from a prospective multicentre cohort registry, the optimised catheter valvular intervention-TAVI registry. Nosotros compared the ii-year cardiovascular and non-cardiovascular bloodshed and in-hospital outcomes between patients with and without preprocedural beta-blocker administration by propensity score matching (PSM).
Results Preprocedural beta blockers were prescribed in 867 patients (33.eight%). After PSM, the incidence of in-hospital congestive heart failure was significantly lower in patients with preprocedural beta blocker (p=0.046). No differences were establish in ii-yr cardiovascular and non-cardiovascular mortality. In the subgroup analyses, beta-blocker administration was associated with a lower cardiovascular bloodshed within two years in patients with a history of coronary avenue featherbed grafting (CABG; log-rank p=0.017), presence of peripheral artery disease (PAD; log-rank p=0.003) and encephalon natriuretic peptide (BNP) ≥400 pg/mL (log-rank p=0.003). When stratified by postprocedural left ventricular ejection fraction (post-LVEF), beta-blocker assistants was associated with a lower cardiovascular mortality among patients with post-LVEF <fifty% (log-rank p=0.024).
Conclusions Preprocedural beta-blocker administration was not associated with two-year cardiovascular and non-cardiovascular bloodshed in overall, simply was associated with a lower 2-year cardiovascular bloodshed in patients with a history of CABG, presence of PAD, BNP ≥400 pg/mL and post-LVEF <50%. The findings must be validated using randomised trials.
- aortic valve disease
- beta blockers
- percutaneous valve therapy
https://creativecommons.org/licenses/past/4.0/
This is an open access article distributed in accordance with the Creative Commons Attribution four.0 Unported (CC Past iv.0) license, which permits others to copy, redistribute, remix, transform and build upon this work for any purpose, provided the original work is properly cited, a link to the licence is given, and indication of whether changes were made. See: https://creativecommons.org/licenses/by/4.0/.
Statistics from Altmetric.com
- aortic valve disease
- beta blockers
- percutaneous valve therapy
Fundamental questions
What is already known most this bailiwick?
-
Transcatheter aortic valve replacement (TAVR) is an established therapy for symptomatic patients with severe aortic stenosis. All the same, the optimal medical therapy, including beta blockers for patients who undergo TAVR, remains unspecified.
What does this report add?
-
The study showed that beta-blocker assistants was associated with a lower 2-year cardiovascular mortality in patients with a history of coronary artery bypass grafting (CABG; log-rank p=0.017), presence of peripheral artery disease (PAD; log-rank p=0.003), brain natriuretic peptide (BNP) ≥400 pg/mL and postprocedural left ventricular ejection fraction (LVEF) <fifty% (log-rank p=0.024).
How might this impact on clinical impact?
-
This result suggests that beta-blocker administration was associated with good clinical outcomes amid patients with a history of CABG, presence of PAD, BNP ≥400 pg/mL and post-procedural LVEF <50%. The findings need to be confirmed by randomised trials.
Introduction
Information technology is not articulate whether medical therapy is advantageous for the treatment of aortic stenosis (AS). Several prior studies have shown that renin–angiotensin system (RAS) inhibitors for As are associated with improved survival.1 ii Information technology is also well known that beta blockers should exist carefully administered in cases of severe AS because of their negative inotropic furnishings. Nonetheless, there are conflicting reports on survival benefits when beta blockers are used in patients with severe As.3 Beta blockers reduce the haemodynamic and metabolic overload in patients with asymptomatic astringent AS.iv
Transcatheter aortic valve replacement (TAVR) is an established therapy in patients with symptomatic severe As at loftier or intermediate surgical take a chance.5–vii Recently, the placement of aortic transcatheter valves 3 trial showed that TAVR with a airship-expandable valve was more effective than surgical aortic valve replacement (SAVR) with respect to the composite endpoint of death, stroke and rehospitalisation at 1 year in patients with severe As at low surgical risk.8 Also, TAVR with a self-expanding valve was not-inferior to SAVR with respect to expiry or disabling stroke in patients at low surgical risk.9 Although the indication of TAVR is expanding based on these studies, there are limited information regarding the optimal medical handling for patients undergoing TAVR. The use of RAS inhibitors after TAVR has lowered mortality.10 xi However, the result of preprocedural beta-blocker assistants on these patients remains unclear. Thus, our purpose was to investigate the effect of preprocedural administration of beta blockers on the clinical outcomes of patients with severe AS who underwent TAVR.
Methods
Study population
A total of 2588 patients were enrolled in the Optimised transCathEter vAlvular iNtervention (Bounding main)-transcatheter aortic valve implantation (TAVI) registry between October 2022 and May 2022. The OCEAN-TAVI registry is a prospective, multicentre, observational registry of patients who underwent TAVR using the Edwards Sapien XT/Sapien three prosthesis (Edwards Lifesciences, Irvine, California, USA) or Medtronic Corevalve/Evolut R prosthesis (Medtronic, Minneapolis, Minnesota, USA) at 14 Japanese medical centres. All study participants provided informed consent and the registry was approved by the ethics committees of all participating institutions. Patients or the public were not involved in the design, or conduct, or reporting, or dissemination plans of our research. The OCEAN-TAVI registry was registered with the University Hospital Medical Information Network Clinical Trial Registry and accepted by the International Commission of Medical Journal Editors (UMIN-ID: 000020423). Patients were followed annually at the participating institutions. At that time, blood tests, echocardiography were performed. The events were site reported from the participating institutions and non adjudicated in clinical event adjudication committee. The database was regularly audited by the data committee members to ensure the consistency.
Afterward exclusion of 25 patients attributable to conversion to open surgery, 2563 patients participated in this report and were divided based on whether or not they were administered preprocedural beta blockers (figure ane). Preprocedural beta-blocker administration was divers every bit beta blockers beingness prescribed at TAVR.
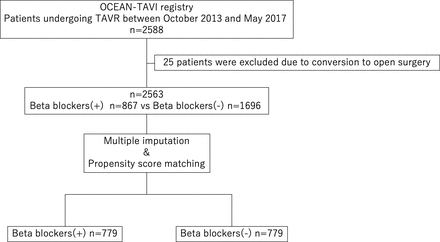
- Download figure
- Open in new tab
- Download powerpoint
Figure ane
Report menses diagram. Of 2588 patients, 25 patients were excluded due to conversion to open surgery. Beta-blocker (−)=patients without preprocedural beta blockers; beta-blocker (+)=patients with preprocedural beta blockers. Body of water, Optimised transCathEter vAlvular iNtervention; TAVR, transcatheter aortic valve replacement.
Outcomes
The primary outcomes were ii-year cardiovascular and non-cardiovascular mortalities later TAVR. For patients who lost to follow-up, we used the last date when survival was confirmed. The secondary outcomes were as follows: in-hospital outcomes and complications and postprocedural echocardiographic information. Cardiovascular mortality and complications were defined based on the Valve Academic Research Consortium (VARC)-2 criteria.12 In-hospital congestive heart failure was defined as the requirement of intravenous injection of diuretics or inotropic agents, mechanical support for center failure, such every bit intra-aortic airship pumping (IABP) or extracorporeal membrane oxygenation (ECMO) after TAVR.
Echocardiography
Transthoracic echocardiography was performed at baseline, before hospital belch, and at the annual follow-upward. All transthoracic echocardiographic parameters were measured according to the guidelines of the American Guild of Echocardiography.13 14 The degree of paravalvular leak (PVL) was measured in accordance with the VARC-2 criteria and reported as a semiquantitative form: none, trace, mild, moderate and severe. Prosthesis-patient mismatch (PPM) was divers as an indexed effective orifice area <0.85 cm2/grandtwo according to the VARC-2 criteria.
Statistical analysis
We compared baseline characteristics between patients with and without preprocedural administration of beta blockers (table 1). Continuous variables are presented as medians and IQRs (25%–75%) and compared using the Educatee's t-test or Mann-Whitney U test. Categorical variables are presented as means and percentages and compared using the Pearson's χ2 examination or Fisher's exact examination.
View this table:
- View inline
Table i
Baseline characteristics
Baseline variables are listed in table ane. In that location were missing data for baseline variables. Percentage of missing data for baseline variables are shown in online supplementary table i. There were no missing data for baseline variables not in online supplementary table 1. Multiple imputation was performed to properly handle missing information. Missing continuous variables were imputed using the predictive mean matching method. Missing binary variables were imputed using logistic regression models. We created xx imputed datasets. Propensity score matching (PSM) was performed later on multiple imputations to reduce imbalances at baseline between the groups. The covariates included in the model are listed in table 1. Propensity scores were calculated within each imputed dataset using logistic regression models to estimate the probability of preprocedural beta-blocker prescription. Propensity scores were averaged across the imputed datasets for each patient. PSM was performed with a ane:one matching protocol without replacement past the averaged propensity scores. The calliper width was 0.2 of the SD of the logit of the averaged propensity scores. Remainder between the two groups was assessed by absolute standardised hateful departure (SMD).
Supplemental material
Cumulative incidences were calculated using the Kaplan-Meier method in the matched accomplice. The log-rank test was performed to compare cardiovascular mortality betwixt patients with and without preprocedural administration of beta blockers.
To identify the groups for which beta blockers were effective, subgroup analyses for cardiovascular bloodshed were performed for historic period (≥80 or <80 years), sex, chronic kidney illness (CKD), atrial fibrillation (AF), coronary artery disease (CAD), coronary artery featherbed grafting (CABG), peripheral artery disease (PAD), preprocedural left ventricular ejection fraction (pre-LVEF; ≥50% or <fifty%), preprocedural aortic regurgitation (AR; ≥moderate or ≤mild), preprocedural mitral regurgitation (MR; ≥moderate or ≤mild) and encephalon natriuretic peptide (BNP; ≥400 pg/mL or <400 pg/mL). In subgroup analyses for cardiovascular mortality, patients with BNP ≥400 pg/mL were focused considering information technology is said that at that place is a loftier possibility of centre failure if BNP ≥400 pg/mL.15 16 In addition, nosotros performed the subgroup analysis for cardiovascular mortality in the subset of PPM and PVL.
To confirm the safety of beta blockers for patients undergoing TAVR, subgroup analyses for non-cardiovascular mortality were performed for age (≥80 or <80 years), sex, body mass alphabetize (≥twenty kg/grand2 or <20 kg/thouii), New York Heart Clan (NYHA) class (1, ii or three, 4), CKD, PAD, chronic obstructive pulmonary disease (COPD), albumin levels (≥3.5 k/dL or <three.five g/dL) and clinical frail score (ane–iv, 5–six or ≥7). Interaction tests between each covariate were performed. PSM after multiple imputation was performed in the subgroups with p for interaction <0.05. We selected the subgroup from the original information including 2563 patients, then performed PSM for preprocedural beta blockers because the baseline characteristic between beta-blocker group and non-beta-blocker grouping would exist consistent in the subgroup. The log-rank test was performed to evaluate cardiovascular mortality between patients with and without preprocedural beta-blocker administration in the subgroups.
It was hypothesised that the effect of beta blockers on cardiovascular mortality would be influenced by LVEF. Therefore, the original information were divided according to pre-LVEF (≥fifty% or <l%), the postprocedural LVEF (post-LVEF; ≥l% or <50%). PSM for preprocedural beta-blocker administration was performed in each group. The log-rank examination was performed to evaluate cardiovascular mortality.
All statistical analyses were performed using SPSS V.23.0 (IBM SPSS Statistics, IBM Corporation) and R software V.3.5.2 (Vienna, Republic of austria). All tests were two sided and statistical significance was set at p<0.05.
Results
Baseline characteristics
The baseline characteristics of the study participants are shown in table ane. Among 2563 patients, beta blockers were administered to 867 patients (33.8%) before TAVR despite the presence of astringent AS (effigy 1). There were statistically significant differences between patients with and without beta-blocker administration with regard to NYHA class 3 or iv, history of ischaemic stroke, CABG, presence of hypertension, CKD, PAD, CAD, AF, implantation of permanent pacemaker, Society of Thoracic Surgeons Predicted Risk of Bloodshed score, Logistic Euro score, Euro Ii score, any diuretic therapy, haemoglobin, estimated glomerular filtration charge per unit, albumin levels, BNP, aortic valve surface area, acme velocity, hateful force per unit area gradient, LV terminate-systolic bore, left atrial diameter, LVEF, indexed stroke book, MR ≥moderate and the transfemoral arroyo.
Chief outcomes in the matched cohort
A full of 1558 patients were matched (online supplementary tabular array 2). After performing PSM, the accented SMD value was lower than 0.1 in all examined covariates.
Kaplan-Meier curves of cardiovascular and non-cardiovascular mortality in the non-matched and matched accomplice are shown in figure ii. Follow-up rate at 2 years was 92.1%. The median follow-up catamenia in the matched cohort was 658 days (IQR 381–863). 90-five patients died of cardiovascular causes and 154 of non-cardiovascular causes during the follow-up menses in the matched accomplice. There was no pregnant divergence between the 2 groups regarding cardiovascular (log-rank p=0.43) and non-cardiovascular mortality (log-rank p=0.29) in the matched cohort.

- Download figure
- Open in new tab
- Download powerpoint
Figure 2
Kaplan-Meier curves of cardiovascular mortality and not-cardiovascular mortality in the non-matched and matched cohort. 2-yr cardiovascular mortality and non-cardiovascular mortality of patients with beta-blocker assistants compared with those without in the (A) non-matched and (B) matched accomplice. TAVR, transcatheter aortic valve replacement.
Secondary outcomes in the matched cohort
Regarding the matched cohort, in-hospital patient outcomes, complications and postprocedural echocardiographic data are shown in tabular array 2. The incidence of in-hospital congestive eye failure was lower among patients treated with beta blockers than among those treated without (p=0.046).
View this table:
- View inline
Table two
In-hospital outcomes and postprocedural echocardiographic data
Subgroup analyses
Subgroup analyses for cardiovascular mortality are shown in effigy 3. The groups with a pregnant departure were history of CABG (p for interaction=0.035), presence of PAD (p for interaction=0.026) and BNP (p for interaction=0.005). Based on this effect, PSM was performed in the groups. Kaplan-Meier curves of cardiovascular mortality in each group are shown in figure 4. Beta-blocker administration was associated with a significantly lower two-year cardiovascular bloodshed in patients with CABG history (log-rank p=0.017), presence of PAD (log-rank p=0.003) and BNP ≥400 pg/mL (log-rank p=0.003). Subgroup analyses for cardiovascular mortality in the subset of PPM and PVL are shown in online supplementary figure 1. There was no interaction between beta blockers and PPM (p for interaction=0.15), beta blocker and PVL (p for interaction=0.34).

- Download figure
- Open in new tab
- Download powerpoint
Figure three
Subgroup analyses for cardiovascular mortality. Beta-blocker (−)=patients without preprocedural beta blockers; beta-blocker (+)=patients with preprocedural beta blockers. Forest plot representing the HRs of cardiovascular mortality in patients with beta-blocker administration compared with patients without, stratified by preprocedural characteristics. AF, atrial fibrillation; AR, aortic regurgitation; BNP, brain natriuretic peptide; CABG, coronary artery bypass grafting; CAD, coronary artery disease; CKD, chronic kidney illness; LVEF, left ventricular ejection fraction; MR, mitral regurgitation; PAD, peripheral artery disease.

- Download figure
- Open in new tab
- Download powerpoint
Figure four
Cardiovascular mortality in subgroups. Kaplan-Meier bend of cardiovascular mortality in patient with and without a history of coronary artery bypass grafting (CABG), with and without presence of peripheral artery affliction (PAD), with brain natriuretic peptide (BNP) <400 pg/mL and ≥400 pg/mL. TAVR, transcatheter aortic valve replacement.
Subgroup analyses for non-cardiovascular mortality are shown in figure 5. Interaction between COPD and beta-blocker administration was noted (p for interaction=0.035).

- Download figure
- Open in new tab
- Download powerpoint
Figure five
Subgroup analyses for non-cardiovascular mortality. Forest plot representing the HRs of non-cardiovascular bloodshed in patients with beta-blocker administration compared with patients without, stratified by preprocedural characteristics. BMI, body mass index; CFS, Clinical Delicate Score; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; NYHA, New York Middle Association; PAD, peripheral artery disease.
Cardiovascular mortality stratified by LVEF
Kaplan-Meier curves of cardiovascular mortality in each grouping (pre-LVEF ≥l% or <50%, post-LVEF ≥50% or <l%) are shown in figure 6. The administration of beta blockers to patients with post-LVEF <50% was associated with a lower 2-yr cardiovascular bloodshed (log-rank p=0.024). In patients with pre-LVEF ≥50%, pre-LVEF <50% and post-LVEF ≥fifty%, at that place was no significant departure in cardiovascular mortality between the groups with or without beta-blocker assistants (log-rank p=0.61 in patients with pre-LVEF ≥50%, log-rank p=0.22 in patients with pre-LVEF <fifty% and log-rank p=0.75 in patients with post-LVEF ≥l%).

- Download effigy
- Open in new tab
- Download powerpoint
Figure 6
Cardiovascular mortality stratified by left ventricular ejection fraction (LVEF). Kaplan-Meier curve of cardiovascular bloodshed in patient with preprocedural LVEF ≥l% or <50% (A), postprocedural LVEF ≥fifty% or <50% (B). TAVR, transcatheter aortic valve replacement.
Word
The optimal medical therapy for patients undergoing TAVR remains unknown. The main findings of our study showed that the assistants of beta blockers to patients undergoing TAVR was associated with a lower incidence of in-hospital congestive heart failure and cardiovascular mortality in specific groups of patients (patients with a history of CABG, presence of PAD, BNP ≥400 pg/mL, and post-LVEF <50%). To our cognition, the present study is the starting time to identify the clan of beta blockers and clinical outcomes subsequently TAVR.
Apart from some trial demonstrations of survival benefits of beta blockers in patients with center failure with reduced ejection fraction (HFrEF),17–19 there are few reports describing the effect of beta blockers on patients undergoing TAVR. An observational study reported that the absenteeism of beta blockers was an independent predictive factor of expiry following TAVR in patients with a history of breast irradiation.20
Two remaining and frequent complications of TAVR are PVL and pacemaker implantation. Assistants of beta blockers is considered harmful in patients with AR because of a longer diastolic period. Additionally, beta blockers suppress atrioventricular conduction. Therefore, there was business organisation that beta-blocker assistants would increment the risk and impact of these complications. Nonetheless, at that place was no significant difference in PVL degree and new permanent pacemaker implantation rate between patients with and without beta-blocker administration in this study. On the reverse, Younis et al reported that beta-blocker discontinuation was associated with an increased risk of high degree AV cake or AF among patients with TAVR.21
In this study, beta blockers were associated with a lowered non-cardiovascular mortality in patients with COPD. It was assumed that beta blockers were contraindicated in patients with COPD because of potential exacerbation. Recently, beta-blocker utilise in patients with COPD was associated with decrease in all-cause mortality and exacerbation charge per unit.22 However, these results should not be overstated and are across the scope of this study.
Blessberger et al reported that at that place was no clear bear witness of the prevention of beta blockers on congestive middle failure after cardiac surgery.23 However, they too reported that perioperative beta-blocker administration reduces the run a risk of ventricular and supraventricular arrhythmias. These findings may have led to a lower incidence of in-hospital congestive middle failure among patients treated with beta blockers than among those not treated with beta blockers.
All specific groups identified in this study are at loftier chance of heart failure or cardiovascular event. Patients undergoing TAVR with a history of CABG, presence of PAD, high BNP levels or LVEF <l% had high mortality rates.24–27 The possible benefits of beta blockers for high-risk patients with middle failure or cardiovascular events could exist a potential caption for the association of a low cardiovascular mortality with beta-blocker administration in these groups of patients. The efficacy of beta blockers in the treatment of ischaemic heart disease and heart failure has been established. Patients with PAD take a high possibility of polyvascular illness, such as CAD and cerebrovascular disease. In improver, Satiroglu et al reported that the lesion of CAD in patients with PAD was often more severe than that in patients without PAD.28 For that reason, beta blockers may be effective on patients with PAD undergoing TAVR. Some prior studies accept shown that beta-blocker therapy was associated with a low risk of mortality in these patients.17–19 29 30
In this study, in that location was no association betwixt the presence and absence of beta-blocker assistants in cardiovascular mortality in patients with pre-LVEF <l% or ≥50%, although beta-blocker administration was associated with a lower cardiovascular mortality among patients with post-LVEF <fifty%. This result may suggest that beta blockers are effective in patients with LVEF <fifty% even later on severe As improvement by TAVR. Patients with LVEF <50% included those with HFrEF and with mid-range HFEF (HFmEF). A recent meta-analysis showed that beta blockers reduced cardiovascular mortality in patients with HFrEF and HFmEF.31
In this study, nosotros establish that beta blockers were not associated with the run a risk of increased complications and non-cardiovascular mortality. Additionally, we identified the association with beta blockers and clinical outcomes. The optimal medical therapy for patients who undergo TAVR would be important because the indications for TAVR are expanding and the number of these patients is increasing. However, this study could not identify the doses and types of beta blockers. Prospective studies are needed to evaluate these problems.
Written report limitations
This study has several limitations. First, information technology was a non-randomised, retrospective study using information from a prospective multicentre cohort registry including a limited number of patients; nevertheless, this cohort is representative of existent-earth do. Further studies with a larger number of patients are ongoing. Second, the definition of in-infirmary congestive heart failure is unique to this registry. Additionally, no data was available on the amounts, types of infusion or drugs used during the procedure that may affect the onset of congestive heart failure. The issue of low incidence of in-hospital congestive eye failure is across the scope of the effect of beta blockers considering the definition includes mechanical support for heart failure, such as IABP and ECMO. In addition, inotropic agents and mechanical support were also used not only for heart failure but also for the cardiogenic shock management. In-hospital congestive middle failure did not include the cases in which inotropic agents or mechanical support was used for other than center failure. However, the reasons for the use of inotropic agents or mechanical support were site reported from the participating institutions. The diagnosis of heart failure was left to the discretion of the clinicians in each participating institution. Third, no information regarding the dose or period of administration, time schedule or type of beta blockers was included. There was not plenty information of medication status at follow-up. In randomised controlled trials, carvedilol, bisoprolol or metoprolol succinate reduce all-cause mortality in patients with HFrEF.17–19 Additionally, the effect of beta blockers is dose dependent.32 Fourth, in that location was no data on the blood force per unit area and heart charge per unit of patients involved in this study. A depression heart rate is associated with skillful prognosis in patients with middle failure,32 and it is non recommended to use beta blockers in patients with hypotension or bradycardia. Lastly, there was a possibility that some of the meaning results were due to the multiple testing. In improver, the number of the events of secondary outcomes and the result in the subgroups was relatively small. The statistical ability may be limited. Therefore, more meticulous studies including larger patients that consider these should be our time to come targets.
Conclusions
Preprocedural beta-blocker assistants for patients undergoing TAVR was not associated with two-year cardiovascular and non-cardiovascular mortality in overall, merely was associated with a lower gamble of ii-twelvemonth cardiovascular mortality simply in patients with a history of CABG, presence of PAD, BNP ≥400 pg/mL and mail-LVEF <50%. These patients may be better prescribed beta blockers. The findings demand to be confirmed by randomised trials.
Acknowledgments
The authors thank all the OCEAN-TAVI investigators.
Request Permissions
If you wish to reuse any or all of this article please utilise the link beneath which will have you lot to the Copyright Clearance Centre's RightsLink service. You will exist able to get a quick price and instant permission to reuse the content in many different ways.
Copyright data:
© Writer(s) (or their employer(s)) 2022. Re-apply permitted nether CC BY. Published past BMJ. https://creativecommons.org/licenses/by/four.0/ This is an open access article distributed in accordance with the Artistic Commons Attribution iv.0 Unported (CC BY iv.0) license, which permits others to re-create, redistribute, remix, transform and build upon this piece of work for any purpose, provided the original work is properly cited, a link to the licence is given, and indication of whether changes were made. Encounter: https://creativecommons.org/licenses/by/4.0/.
Source: https://openheart.bmj.com/content/7/2/e001269
Posted by: benedicttherem.blogspot.com


0 Response to "Do You Have To Take Beta Blockers After Valve Repair"
Post a Comment